It is expressed as a multiple of one-twelfth the mass of the carbon-12 atom, 1. 
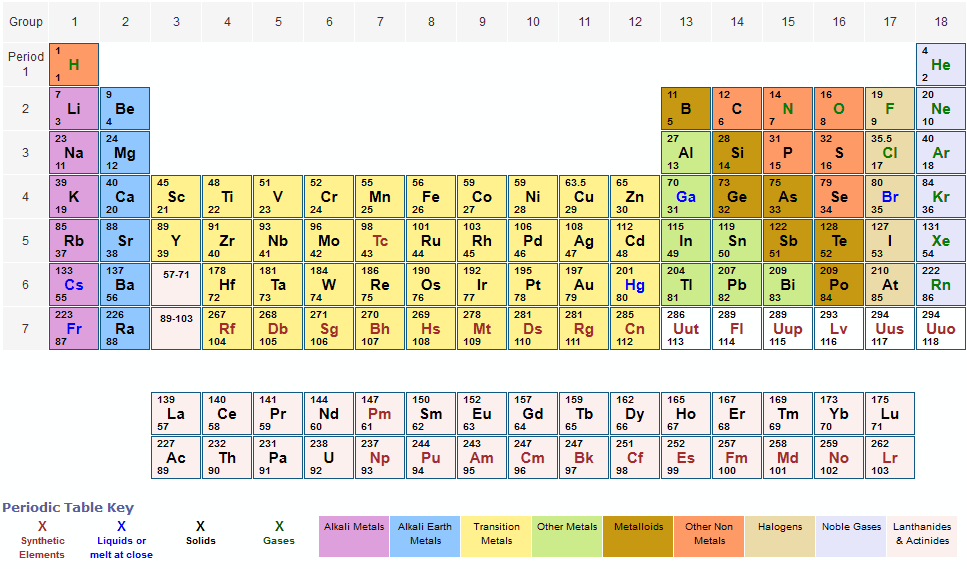
You will need to refer to a periodic table for proton values. atomic mass, the quantity of matter contained in an atom of an element. In this notation, the atomic number is not included. Click on Element Atomic Number, Element Symbol, Element Name and Element Atomic Weight headers to sort.Element Atomic Number Element Symbol Element Name Element Atomic Weight 1: H: Hydrogen: 1.00794: 2: He: Helium: 4.002602: 3: Li: Lithium: 6.941. Symbol-mass format for the above atom would be written as Cr-52. The atomic weight Ar(E) of element E is defined by the International Union of Pure and Applied Chemistry (IUPAC) as the relative molar mass 12M(E)/M(12C). Atomic Weight table gives the Atomic Weight of all the elements of periodic table. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. The "A" value is written as a superscript while the "Z" value is written as a subscript. Both the atomic number and mass are written to the left of the chemical symbol. Spontaneous formation of junctions and rings via self-organization. The composition of any atom can be illustrated with a shorthand notation called A/Z format. Engineering self-integrated atomic quantum wires to form nano-networks. In 1869, Dmitri Mendeleev and Lothar Meyer individually came up with their own periodic law 'when the elements are arranged in order of increasing atomic mass, certain sets of properties recur periodically.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
